In this article, we will look deeper into the concept of what is medical technology, how the Medtech market is being transformed recently, and look for the forecasts.
MedTech definition
Can you imagine the patient’s life quality improvement without MedTech products? Certainly not. Hence, every device, service, or solution that is using medical technology to take care of people’s health, either by standard precautions, diagnosing, monitoring, or the very treating is medical technology or MedTech.
It makes sense to compare medical technologists to the crew members of a theater performance. Unlike actors, they work backstage never coming out for applauds and flowers. In the hospital, patients will hardly see a medical technologist among doctors and nurses workflow but what they do to healthcare is truly vital.
MedTech is so common today and so tightly it became an important part of our daily life that living without this technology seems surreal.
Medical device technology, for instance, in particular, its early diagnostic tools can be used for disease prevention, and the early diagnosis provides successful treatment in time.
In our current COVID-19 pandemic reality MedTech services have been saving thousands of lives via digital health, medical, and in-vitro diagnostic devices providing fast testing equipment, respirators, and sharing information on how to stay safe, keep sane and avoid the spread of coronavirus.
On the MedTech market
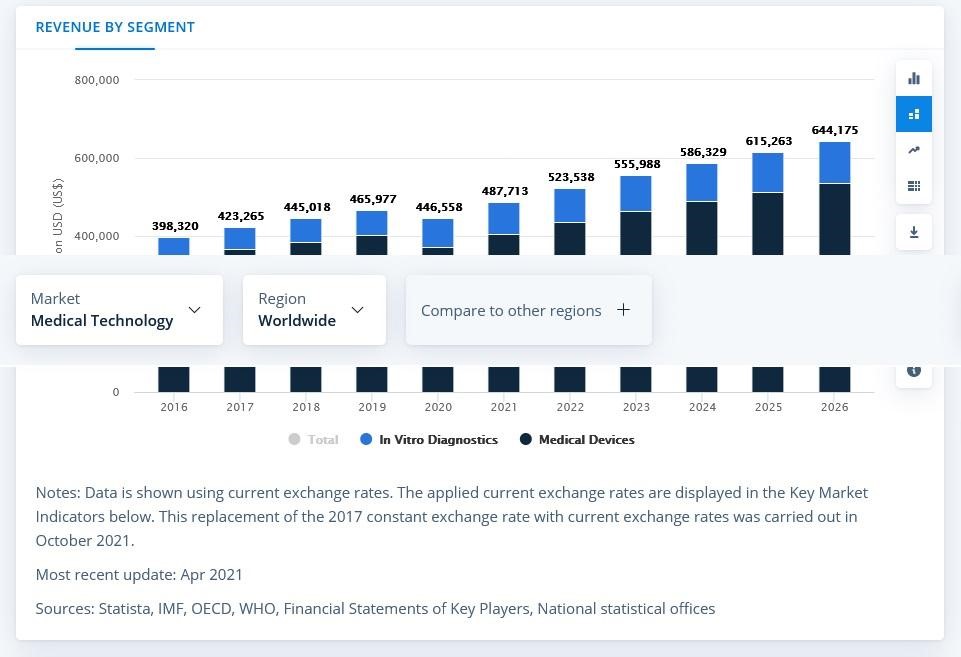
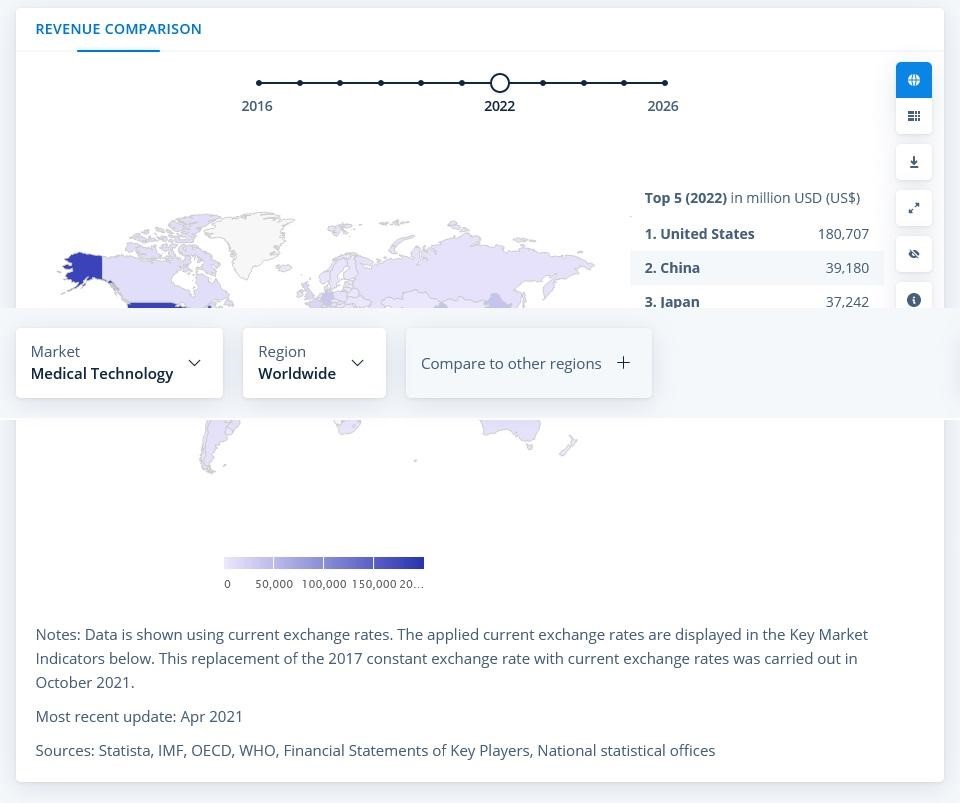
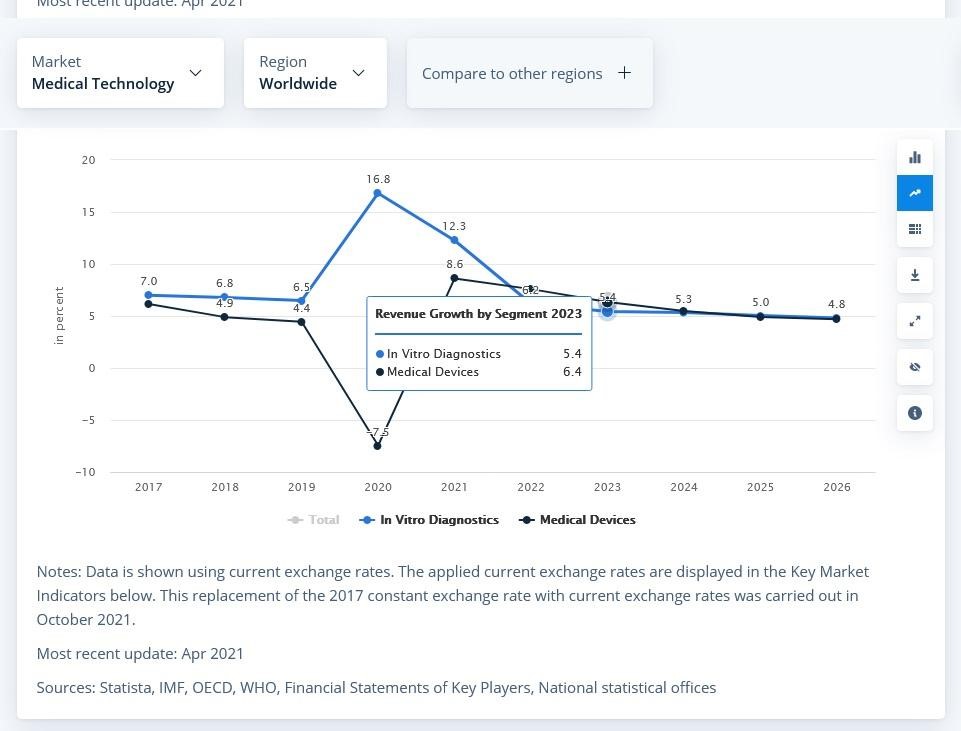
According to statista.com, the Medical Technology market was estimated at almost 488 million USD in 2021. Medical Devices part was the biggest with 406 million USD and the annual growth rate is forecasted to be 5.32%, with the market volume of 644 million USD by 2026, as a result. In-vitro diagnostic devices are regarded to remain the number one for the upcoming years.
Talking about Europe, its MedTech market is extremely rewarding and engaging to join. Yet, as we all know how tough the European Union is in terms of high standards, it takes effort to face all the administrative requirements. That is why a detailed investigation and study of all that is needed to get CE-marking and certification are necessary.
Moreover, to obtain your medical device technology approval, you have to be aware of all the important supplementary rules like standards for software, risk analysis, or norms on recording, designing, or transmitting a clinical investigation with MedTech services.
What Do Medical Technologists do?
Scientifically correct and precise data is what medical technologists are responsible for. It is the very foundation of the right diagnosis and effective treatment. No wonder the hospital laboratory professionals working on various tests are often called med lab scientists.
After earning their degree from a four-year university study, medical technologists can operate complex equipment, that costs millions of dollars. From simple blood tests to monitoring pregnancy complications, drug therapy, or complex testing that uncovers serious diseases, it is due to their skills the doctors constantly depend on.
In the Chemistry area, medical technologists’ main concern is proteins and electrolytes. This is where the patient finds out about the sugar level in his blood, whether he has an allergy to antibiotics, etc. The medical technologist’s focus in Microbiology is bacteria, fungi, and parasites. In Blood banking, they define types of blood and prepare donor units. Urinalysis shows a lot about our health conditions like infections, kidney damage, as well as pregnancy. The professionals in Hematology and Coagulation perform the Complete Blood Count (CBC) tests determining the numbers of white and red blood cells and measuring hemoglobin.
Point-of-care solutions market
Being in the middle of the transformation process, the healthcare industry is going through reform via many initiatives. The priorities go to implementing electronic patient data, tying provider compensation to high-quality metrics instead of usual bills for services, developing responsible care institutions, and making patient outcomes better in general.
Among the main facilitators of these changes is the increase of point-of-care solutions. Promising low costs through easier access to and use of diagnostic and monitoring devices POC tech guarantees much better outcomes.
Above all, simple point of care devices are supposed to be used at the basic level of healthcare and remotely with no laboratory foundation needed. In the comfort of their homes patients are able to do self-tests. It is a great relief for stigmatized issues such as AIDS and other sexually transmitted illnesses where people feel disgraced and socially discriminated against.
With the help of POC technologies, the care of numerous conditions and diseases is greatly improved, mainly when we deal with few resources within a poor healthcare infrastructure where professional long-term medical service is a fortune. The new kind of test provides fast and accurate results, making it possible for starting the right therapy in time as well as other healthcare support.
The point of care market is diverse and it embraces a vast range of products both for patients and medical technologists, either within a traditional healthcare institution like a hospital or laboratory or remotely.
It will take time
Industry experts point pot that it will take some time for consumer markets to blossom in their full power. The point of care solutions will definitely have to be proven in traditional settings before both the consumers and regulators feel confident and safe for implementing on a large scale.
It is true that a few segments of the POC markets like fitness and sports have evolved immensely over the previous years. Yet most of the monitoring and diagnostics point-of-care apps for clinical indications such as infections and cardiology are in their infancy when it comes to serious markets.
Sooner or later, the POC consumer market will become a powerful source of income for the MedTech devices industry. We only have to be patient to wait at least 5-10 years or for special indications even more. Being realistic and reasonable in this matter is important and in the long term perspective very much rewarding.
Different technologies – different prices
Novel imaging and electrodes-based solutions tend to lend themselves to more focused and guided application areas, as they usually require highly skilled operators and medical technologists to read and explain the results.
On the other hand, In vitro diagnostic (IVD) solutions will find their way far down the healthcare settings continuum for all indication areas. A steady stream of solutions is expected to appear either from large corporations or small startups intended at addressing, for instance, circulating tumor cell detection done in the physician’s cabinet or rapid scrub typhus screening in poor districts of India.
It is inevitable that the advances in data analytics and algorithms will eventually technically make the use of these devices outside of hospitals and clinics possible. However, a longer time will be needed for regulators to validate complex image interpretation-based diagnostics and monitoring solutions for use in homes and nursing houses.
Redefining priorities
The medical technology industry is being offered immense opportunities due to constant great innovations in analytics and AI. Yet, these go hand in hand with costs covering the whole industry, margin tensions, new ways of payment and making contracts, increased regulatory requirements worldwide, and other shifts and changes in the markets.
The leading Medtech companies are reconsidering their top preferences and developing business models for more opportunities. They do efforts in facing challenges whether it is referred to legal, commercial, technical areas, etc.
At ZenBit, we know that for the MedTech industry, you have no choice but to take up transformation for your business success and its future flourishing. ZenBeat team offers a rich array of MedTech services and solutions created to help healthcare companies worldwide.

Conclusion
Let our team help you with your product quality and regulatory compliance. Our developers have a diligent work ethic and a great interest in problem-solving to provide services that drive solutions that make a difference. Contact for your medical technology consulting services now! Together we will define effective strategies for your business needs!






